Acrylamide
|
|
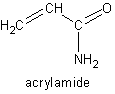
The chemical compound acrylamide is an amide with chemical formula C3H5NO and structure
It is a white odorless crystalline, soluble in water, ethanol and ether. It is used in papermaking, ore processing, manufacture of permanent-press fabrics, and the manufacture of dyes. Acrylamide is also used to synthesize polyacrylamides which find many uses as water-soluble stabilizers, for instance in waste water treatment and in gel electrophoresis (SDS-PAGE).
The CAS registry number for acrylamide is 79-06-1 and its UN number is 2074.
In April 24 2002, swedish Livsmedelsverket (National Food Administration) alarmed that acrylamide may be a carcinogen, and concern was raised as it can be found in small quantities in baked and cooked foods, such as potato chips and breads. It is believed to be created in foods when starches and other carbohydrates are overheated far past 100°C. Some sources claim that people in the modern world consume daily on average around 25 micrograms of acrylamide, and that this accounts for a significant number of cases of cancer.
However, since then, further research has not revealed any connection between acrylamide intake via food and cancer in the large intestine, rectum, bladder, or kidneys. And in 2005, in a cooperation between Harvard School of Public Health and swedish Karolinska Institutet, researchers were seeking a connection between acrylamide and breast cancer, but could not find any in that case either, as revealed in a press release published March 15, 2005. The current opinion among most researchers is therefore that former acrylamide reports have been exaggerated by media, although with the caveat that any substance in "astronomical doses" can still be dangerous to any organism, including humans. However, in the case of acrylamide, such doses are currently believed to be far greater than the acrylamide intake from food. Unfortunately, warnings by media had sometimes been misunderstood. For example, there were earlier reports of 200-700 swedes dying annually due to acrylamide intake via food; however these results were in fact based on lab rats.
However, acrylamide may still be a carcinogen if exposed to it in larger doses. There are limited evidence large doses can cause damage to the male reproductive glands. Direct exposure to pure acrylamide by inhalation, skin absorption, or eye contact irritates the exposed mucous membranes, e.g. the nose, and can also cause sweating, urinary incontinence, nausea, myalgia, speech disorders, numbness, paresthesia, and weakened legs and hands.
External links
- http://www.nsc.org/library/chemical/Acrylami.htm Chemical information
- http://www.konsumentverket.se/html-sidor/livsmedelsverket/enginformationakryl.htm Report about acrylamide in food and cancer risks
- http://www.crime.co.nz/c-files.asp?ID=46 Court case about a suspected acrylamide poisoning
- http://www.hsph.harvard.edu/press/releases/press01282003.html Harvard School of Public Health press release; baked and fried food does not increase risk of certain cancers in humans
- http://www.hsph.harvard.edu/press/releases/press03152005.html Harvard School of Public Health press release; no breast cancer risks from acrylamide via foodde:Acrylamid