Alkyl
|
|
An Alkyl is a univalent radical containing only carbon and hydrogen atoms arranged in a chain. The alkyls form a homologous series with the general formula CnH2n+1. Examples include methyl, CH3· (derived from methane) and butyl C2H5· (derived from butane). They are normally not found on their own but are found as part of larger branched chain organic molecules. On their own they are free radicals and therefore extremely reactive.
| Contents |
Structure
The structure of an alkyl is the same as its straight chained alkane counterpart but with one less hydrogen atom. For example, this is the structure of Methyl, the smallest alkyl:
Missing image
Alkyl.png
Image:Alkyl.png
Alkyls are radicals since only three out of four valency electrons are used in bonding on one of the carbon atoms.
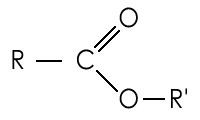
Since they are radicals, alkyls are normally found as part of larger structures. Radicals are represented in general formulae using the shorthand symbol R. This is shown in the general displayed formula of an ester below:
In many cases R represents an alkyl, although it may represent any radical.
Reactions
Unbonded alkyls are free radicals, therefore they are formed only as instantaneous intermediates in reactions usually involving other free radicals. When a free radical is formed it very quickly reacts with something else to lose the unpaired electron.
An example of a reaction where alkyls are formed instaneously is chlorination. This reaction involves chlorine free radicals, formed by photodissociation, reacting with an an organic molecule, ussually an alkane. The chlorine free radical will bond with a hydrogen from an alkane forming hydrochloric acid and leaving an alkyl. This alkyl will then bond to a chlorine to form the product; a chloroalkane. The reaction is very energetic and can be explosive if proper precautions are not taken. This is because of the reactivity of free radicals.
Naming Alkyls
The naming convention for alkyls is much the same as alkanes. The suffix is always -yl. The prefix depends on how many carbon atoms are in the molecule. This uses the same system as for Alkanes as shown in this table (taken from IUPAC nomenclature):
| Number of carbons | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| Prefix | Meth | Eth | Prop | But | Pent | Hex | Hept | Oct | Non | Dec | Undec | Dodec |
For example, the first three alkyls are called Methyl, Ethyl and Propyl.
These names are used to name branched chained structures, for example 3-MethylPentane:
Missing image
3-MethylPentaneHighlighted.png
Image:3-MethylPentaneHighlighted.png
The structure of 3-MethylPentane is made of two parts. First, the longest straight chain of Carbon atoms; 5 Carbons long with no unsaturated bonds, thus named Pentane (Highlighted Blue). Second, there is an Alkyl group; Methyl (Highlighted Red). Alkyl groups are included in the name of a molecule before anything else. Thus the name becomes MethylPentane. This name is, however, ambiguous, as the Methyl branch could be on any one of three different carbon atoms (Second, Third or Fourth, from left to right). Thus the name becomes 3-MethylPentane to avoid ambiguation.
If there is more than one of the same Alkyl group attached to a chain, then the name becomes slightly different, for example 2,3,3-triMethylPentane:
Missing image
2,3,3triMethylPentane.png
image:2,3,3triMethylPentane.png
Here there are three identical alkyl groups attached to carbon atoms 2, 3 and 3. There name includes 2,3,3 to avoid ambiguation on the position of the groups and 'tri' to indicate that there are three identical alkyl groups, in this case they are all methyls. Suppose the methyl attached to carbon atom no.2 was, in fact, an Ethyl. The name would become 2-Ethyl-3,3-diMethylPentane. Note that when there are different alkyl groups, they are listed in alphabetical order.