Rate of enzyme mediated reactions
|
|
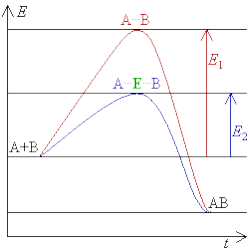
The rate of enzyme mediated reactions is the rate of chemical reactions mediated by enzymes. Enzymes can increase reaction rate by favoring or enabling a different reaction pathway with a lower activation energy, making it easier for the reaction to occur.
| Contents |
|
|
Factors influencing rate
The overall rate of enzyme mediated reactions depends on many factors including properties of the cellular environment, modifications to the enzyme and inhibition.
Cellular environment
- Temperature All chemical reactions speed up as temperature is raised. Extremes of temperature can denature an enzyme so that it can no longer function. The temperature at which the enzyme exhibits maximum activity is called the enzyme's temperature. Temperatures around 40-50°C denature most proteins.
- Substrate concentration
- Enzyme concentration De novo synthesis (the production of more enzyme molecules) increases catalysis rates.
- Enzyme activity Enzyme activity is the catalytic effect exerted by an enzyme.
- pH Extremes of pH can denature an enzyme so that it can no longer function. Many enzymes function optimally in the neutral pH region. There are exceptions; pepsin, a stomach enzyme, functions only in very acidic conditions, and so cannot work in the small intestine.
- Salt concentration Extremes of salt concentration can inactivate an enzyme.
Modifications
- Posttranslational modification: More specific regulation of reaction rate is possible by posttranslational modification (e.g., phosphorylation) of the enzyme or by cofactors like metal ions or organic molecules (e.g., NAD+, FAD, CoA, or certain vitamins) that interact with the enzyme.
- Allosteric modulation: Allosteric enzymes have either effector binding sites, or multiple protein subunits that interact with each other and thus influence catalytic activity.
Inhibition
Enzymes reaction rates can be changed by competitive inhibition, non-competitive inhibition, uncompetitive inhibition and mixed inhibition.
Competitive inhibition
The inhibitor may bind to the substrate binding site as shown in the figure, thus preventing substrate binding. Alternatively, binding of substrate to a different binding site may change protein conformation in a way that prevents substrate binding, while substrate binding changes protein conformation to prevent inhibitor binding. These two possibilities can not be distinguished by enzyme kinetics (X-ray crystallography would be a suitable method), thus the frequently found statement that competitive kinetics proves inhibitor binding at the substrate site is false.
Uncompetitive inhibition
Uncompetitive inhibition occurs when the inhibitor binds only to the enzyme-substrate complex, not to the free enzyme, the EIS complex is catalytically inactive. This mode of inhibition is rare.
Non-competitive inhibition
Non-competitive inhibitors never bind to the active center, but to other parts of the enzyme that can be far away from the substrate binding site. By changing the conformation (the three-dimensional structure) of the enzyme, they disable or enable the ability of the enzyme to turn over its substrate, while substrate binding is still possible (in other words: there is no competition between substrate and inhibitor for the enzyme). The EIS-complex has no catalytic activity.
Partially competitive inhibition
The mechanism of partially competitive is similar to that of non-competitive inhibition, except that the EIS-complex has catalytic activity, which may be lower or even higher (partially competitive activation) than that of the ES-complex.
Enzyme inactivation
Inactivators bind to the enzyme irreversibly (while binding of inhibitors is per definitionem reversible). Of particular medical importance are suicide inactivators which bind to the enzyme at a substrate binding site and are then turned over into a compound and do not release the site, preventing the substrate from binding. Certain irreversible neurotoxins, for example, have this property, while other, reversible, toxins do not.
Kinetics
Enzyme kinetics is referred to as Henri-Michaelis-Menten kinetics. The Michaelis constant, and the Lineweaver-Burke diagram help to define enzyme kinetics.