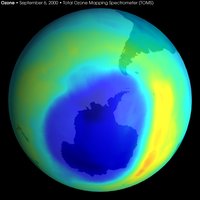
Ozone depletion
|
|
Ozone depletion refers to the phenomenon of reductions in the amount of ozone in the stratosphere. There was a reduction of approximately 5% detected from 1979 to 1990. Since the ozone layer prevents most harmful wavelengths of ultraviolet light from passing through the Earth's atmosphere, observed and projected decreases in ozone have generated worldwide concern and led to adoption of the Montreal Protocol banning the use of chlorofluorocarbon (CFC) compounds, as well as other ozone depleting chemicals such as carbon tetrachloride, trichloroethane (also known as methyl chloroform), and bromine compounds known as halons.
Ozone depletion varies geographically and by season. The term ozone hole refers to the annual, temporary reductions in the polar regions, where large losses in ozone occur each spring (up to 70% over 25 million km2 of Antarctica and 30% over the Arctic) followed by recovery in the summer. This reduction is caused by an increase in concentrations of stratospheric chlorine from breakdown of human manufactured CFC emissions, as well as other gases.
In public policy discussions, the term ozone layer depletion is considered synonymous with the theory that a trend of global ozone depletion, which is mainly caused by CFC emissions, is subsequently allowing more ultraviolet radiation to reach the Earth's surface.
It is suspected that a variety of biological consequences, including, for example, increases in melanoma and the destruction of plankton populations in the ocean's photic zone, may result from the increased UV exposure due to ozone depletion.
Ozone_hole_area.jpg
| Contents |
History of the research
In 1970 Prof. Paul Crutzen pointed out the possibility that nitrogen oxides from fertilizers and supersonic aircraft might deplete the ozone layer.
In 1974 Frank Sherwood Rowland and Mario J. Molina realised that CFCs, like other gases, act as very efficient catalyzers for the breakup of ozone molecules.
James Lovelock (most popularly known as the creator of the Gaia hypothesis) had already discovered, during a cruise in the South Atlantic in 1971, that almost all of the CFC compounds manufactured since their invention in 1930 were still present in the atmosphere.
Crutzen, Rowland and Molina received the Nobel Prize in Chemistry in 1995 for their work. Based on their work, scientists calculated that if CFC production continued to increase at the going rate of 10% per year until 1990 and then remain steady, CFCs would cause a global ozone loss of 5 to 7% by 1995, and a 30 to 50% loss by 2050. However, the discovery of the Antarctic "ozone hole" by Farman, Gardiner and Shanklin (announced in a paper in Nature in May 1985) was a surprise. It was found that chemical reactions on polar stratospheric clouds (PSCs) in the cold Antarctic stratosphere caused faster depletion than expected, which attracted worldwide publicity.
Satellite measurements showing massive depletion of ozone around the south pole were becoming available at the same time. However, these were initially rejected as unreasonable by data quality control algorithms (they were filtered out as errors since the values were unexpectedly low); the ozone hole was only detected in satellite data when the raw data was reprocessed following evidence of ozone thinning in in situ observations.
Ozone depletion has been observed all over the globe but is greatest at high latitudes (that is, near the poles). The best known example is the annual thinning of the ozone layer over Antarctica during the polar spring (see ozone hole section below).
Since 1981 the UNEP has sponsored a series of reports on scientific assessment of ozone depletion. The most recent is from 2002.
Ozone cycle overview
Ozone creation
Ozone in the Earth's atmosphere is generally created by ultraviolet light striking oxygen molecules, which consist of two oxygen atoms (O2), creating two single oxygen atoms, known as atomic oxygen. The atomic oxygen then combines with a molecule of O2 to create ozone, O3. The ozone molecule is rather reactive, and when hit by ultraviolet light it splits again into a molecule of O2 and an atom of atomic oxygen, a continuing process called the ozone-oxygen cycle. Prior to the beginning of the depletion trend, the amount of ozone in the stratosphere was kept even by a stable balance in the amount of creation and destruction of ozone molecules by UV light.
Ozone destruction
Chemical factors
Ozone can be destroyed by a number of free radical catalysts, of which the most important are hydroxyl (OH), nitric oxide (NO) and atomic chlorine (Cl) and bromine (Br). All of these radicals have both natural and anthropogenic (manmade) sources. At the present time, most of the OH and NO in the stratosphere is of natural origin, but human activity has dramatically increased the chlorine and bromine. These elements are found in certain stable compounds, especially chlorofluorocarbons (CFCs), which may find their way to the stratosphere and there be liberated by the action of ultraviolet light. Most importantly, the chlorine atoms so generated destroy ozone molecules in a catalytic cycle. In this cycle, a chlorine atom reacts with an ozone molecule, taking an oxygen atom with it (forming ClO) and leaving a normal oxygen molecule. A free oxygen atom then takes away the oxygen from the ClO, and the final result is an oxygen molecule and a chlorine atom, which then reinitiates the cycle. A single chlorine atom would keep on destroying ozone forever were it not for reactions that remove them from this cycle by forming reservoir species such as hydrochloric acid and chlorine nitrate. On a per atom basis, bromine is even more efficient than chlorine at destroying ozone, but there is much less bromine in the atmosphere at present. As a result, both chlorine and bromine contribute significantly to the overall ozone depletion.
Polar stratospheric clouds
The reactivation of atomic chlorine from these reservoir species is normally slow, but is enhanced by the presence of polar stratospheric clouds which appear during Antarctic winters, leading to a strong seasonal cycle in ozone hole formation.
Observations
Ozone_time_series.jpg
The most pronounced decrease in ozone has been in the lower stratosphere. However, the ozone hole is most usually measured not in terms of ozone concentrations at these levels (which are typically of a few parts per million) but by reduction in the total column ozone, above a point on the Earth's surface, which is normally expressed in Dobson units. Marked decreases in column ozone in the Antarctic spring and early summer compared to the early 1970s and before have been observed using instruments such as the Total Ozone Mapping Spectrometer (TOMS) [1] (http://www.atm.ch.cam.ac.uk/tour/part2.html).
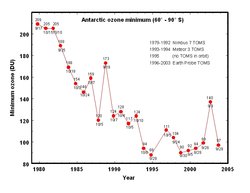
Substantial reductions of up to 70% in the ozone column observed in the austral (i.e., southern hemispheric) spring over Antarctica and first reported in 1985 (Farman et al 1985) are continuing [2] (http://www.wmo.ch/web/arep/reports/o3_assess_rep_2002_front_page.html). Through the 1990's, total column ozone in September and October have continued to be 40-50% lower than pre-ozone-hole values. In the arctic, declines are greatest in winter/spring and the amount is more variable year-to-year than in the Antarctic: when the stratosphere is colder the losses are greater, up to 30%.
The importance of the reaction on polar stratospheric cloud is many-fold. The PSCs only form in extreme cold; the Antarctic stratosphere is colder than the Arctic; which is why ozone holes first formed over Antarctica and why the Arctic ones are not as deep. The original predictions failed to take PSCs into account, which is why a sudden Antarctic ozone hole, rather than gradual global depletion, was such a surprise.
In middle latitudes it is probably preferable to speak of ozone depletion rather than holes; declines are about 3% below pre-1980 values for 35-60N and about 6% for 35-60S. In the tropics, there are no significant trends.
Ozone depletion also explains much of the observed reduction in stratospheric and upper tropospheric temperatures [3] (http://www.grida.no/climate/ipcc_tar/wg1/223.htm) [4] (http://www.giss.nasa.gov/edu/gwdebate/). This is because the reason for the warmth of the stratosphere is absorption of UV radiation by ozone, hence reduced ozone leads to cooling. Some stratospheric cooling is also predicted from increases in greenhouse gases such as CO2; however the ozone-induced cooling is found to be probably dominant.
Predictions of ozone remains a difficult science. The World Meteorological Organization Global Ozone Research and Monitoring Project - Report No. 44 (http://www.al.noaa.gov/WWWHD/Pubdocs/Assessment98/executive-summary.html#A), which on balance comes out strongly in favour of the Montreal protocol, nonetheless notes that projections of ozone loss for the 1994-1997 period made in the UNEP 1994 Assessment had been an overestimate.
Chemicals in the atmosphere
CFCs in the atmosphere
CFCs are completely artificial (they did not exist in nature before being synthesized by man). They are used in air conditioning/cooling units, as aerosol spray propellants prior to the 1980s, and in the cleaning processes of delicate electronic equipment, and are a byproduct of some chemical processes. As mentioned in the ozone cycle overview above, when such ozone-depleting chemicals reach the stratosphere, they are dissociated by ultraviolet light to release chlorine atoms. The chlorine atom acts as a catalyst which can break down many thousands of ozone molecules before it is removed from the stratosphere. Given the longevity of CFC molecules, recovery times are measured in decades. It is calculated that a CFC molecule takes an average of 15 years to go from the ground level up to the upper atmosphere, and it can stay there for about a century, destroying up to one hundred thousand ozone molecules during that time.
Verification of observations
Scientists have increasingly been able to attribute the observed ozone depletion to the increase of anthropogenic halogen compounds from CFCs by the use of complex chemical transport models and their validation against observational data (e.g. SLIMCAT (http://www.lec.leeds.ac.uk/~martyn/slimcat.html)). These models work by combining satellite measurements of chemical concentrations and meteorological fields with chemical reaction rate constants obtained in lab experiments, and are able to identify not only the key chemical reactions but also the transport processes which bring CFC photolysis products into contact with ozone.
The ozone hole and its causes
The Antarctic ozone hole is an area of the antarctic stratosphere in which the recent ozone levels have dropped to as low as 33% of their pre-1975 values. The ozone hole occurs during the Antarctic spring, from September to early December, as strong westerly winds start to circulate around the continent and create an atmospheric container. Within this "polar vortex", over 50% of the lower stratospheric ozone is destroyed during the antarctic spring.[5] (http://www.nas.nasa.gov/About/Education/Ozone/antarctic.html)
Polar regions get a much larger variation in sunlight than anywhere else, and during the 3 months of winter spend most of time in the dark without solar radiation. Temperatures hover around or below -80'C for much of the winter, which leads to the formation of polar stratospheric clouds (PSCs). The cloud particles are composed of nitric acid (Type I PSC) or water ice (Type II PSC); both types provide surfaces for chemical reactions that speed the degradation of ozone molecules.
As explained above, the overall cause of ozone depletion, in the antarctic and elsewhere, is the presence of chlorine-containing source gases (primarily CFCs and related halocarbons) which in the presence of UV light are dissociated releasing chlorine atoms which go on to catalyze the destruction of ozone. The Cl-catalyzed ozone depletion can take place in the gas phase, but it is dramatically enhanced in the presence of polar stratospheric clouds. The photochemical processes involved are complex but well understood. The key observation is that ordinarily, most of the chlorine in the stratosphere resides in stable "reservoir" compounds, primarily hydrogen chloride (HCl) and chlorine nitrate (ClONO2). During the antarctic winter and spring, however, reactions on the surface of the cloud particles convert these reservoir compounds back into reactive free radicals, Cl and ClO. The clouds can also remove NO2 from the atmosphere by converting it to nitric acid, preventing the newly formed ClO from being converted back into ClONO2. The role of sunlight in ozone depletion is the reason why the Antarctic ozone depletion is greatest during spring; during winter, even though PSCs are at their most abundant, there is no light over the pole to drive the chemical reactions. Most of the ozone that is destroyed is in the lower stratosphere, in contrast to the much smaller ozone depletion through homogeneous gas phase reactions, which occurs primarily in the upper stratosphere.
Warming temperatures near the end of spring break up the vortex around mid-December. As warm, ozone-rich air flows in from lower latitudes, the PSCs are destroyed, the ozone depletion process shuts down, and the ozone hole heals.
Interest in ozone hole
While the effect of the Antarctic hole in decreasing the global ozone is relatively small, estimated at about 4% per decade, the hole has generated a great deal of interest because:
- The decrease in the ozone layer was predicted in the early 1980's to be roughly 7% over a sixty-year period.
- The sudden recognition in 1985 that there was a substantial "hole" was widely reported in the press. The especially rapid ozone depletion in Antarctica had previously been dismissed as measurement error.
- Many were worried that ozone holes might start to appear over other areas of the globe but to date the only other large-scale depletion is a smaller ozone "dimple" observed during the Arctic spring over the North Pole. Ozone at middle latitudes has declined, but to a much smaller extent (about 4-5 percent.)
- If conditions became more severe (cooler stratospheric temperatures, more stratospheric clouds, more active chlorine) then global ozone may decrease at a much greater pace. Standard global warming theory predicts that the stratosphere will cool.
- When the Antarctic ozone hole does break up, the ozone-depleted air drifts out into nearby areas. Decreases in the ozone level of up to 10% have been reported in New Zealand in the month following the break-up of the Antarctic ozone hole.
Consequences of ozone depletion
Since the ozone layer absorbs ultraviolet light from the Sun, ozone layer depletion is expected to increase surface UV levels, which could lead to damage, including increases in skin cancer. This was the reason for the Montreal Protocol. Although decreases in stratospheric ozone are well-tied to CFCs, and there are good theoretical reasons to believe that decreases in ozone will lead to increases in surface UV, there is not much direct observational evidence linking ozone depletion to higher incidence of skin cancer in human beings.
Increased UV due to the ozone hole
Ozone, while a minority constituent in the earth's atmosphere, is responsible for most of the main absorption of ultraviolet radiation. The amount of UV radiation that penetrates through the ozone layer decreases exponentially with the thickness/density of the layer. Correspondingly, a decrease in atmospheric ozone is expected to give rise to significantly increased levels of UV near the surface.
Increases in surface UV due to the ozone hole can be partially inferred by radiative transfer model calculations, but cannot be calculated from direct measurements because of the lack of reliable historical (pre-ozone-hole) surface UV data, although more recent surface UV observation measurement programmes exist (e.g. at Lauder, New Zealand [6] (http://www.niwa.co.nz/services/uvozone/)).
Because it is this same UV radiation that creates the ozone in the ozone layer from O2 (regular oxygen) in the first place, a reduction in stratospheric ozone would actually tend to increase photochemical production of ozone at lower levels (in the troposphere), although the overall observed trends in total column ozone are still a decrease, largely because ozone produced lower down has a naturally shorter photochemical lifetime, so it is destroyed before the concentrations could reach a level which would compensate for the ozone reduction higher up.
Biological effects of increased UV
The main public concern regarding the ozone hole has been the effects of surface UV on human health. As the ozone hole over Antarctica has in some instances grown so large as to reach southern parts of Australia and New Zealand, environmentalists have been concerned that the increase in surface UV could be significant.
UVB (the higher energy UV radiation absorbed by ozone) is generally accepted to be a contributory factor to malignant melanoma (skin cancer). One study showed that a 10% increase in UVB radiation was associated with a 19% increase in melanomas for men and 16% for women (Fears et al, Cancer Res. 2002, 62(14):3992-6). Another study of people in Punta Arenas, the southernmost city in the world (53°S), showed a 66% increase in skin cancer in seven years along with with decreased ozone and increased UVB levels (Abarca, Jaime F. & Casiccia, Claudio C. (2002) Skin cancer and ultraviolet-B radiation under the Antarctic ozone hole: southern Chile, 1987-2000. Photodermatology, Photoimmunology & Photomedicine 18 (6), 294-302 [7] (http://www.blackwell-synergy.com/links/doi/10.1034/j.1600-0781.2002.02782.x/full/)).
So far, ozone depletion in most locations has been typically a few percent. Were the high levels of depletion seen in the ozone hole ever to be common across the globe, the effects could be substantially more dramatic. For example, recent research [8] (http://sci.newsfactor.com/perl/story/15776.html) has analyzed a widespread extinction of plankton 2 million years ago that coincided with a nearby supernova. Researchers speculate that the extinction was caused by a significant weakening of the ozone layer at that time when the radiation from the supernova produced nitrogen oxides that catalyzed the destruction of ozone (plankton are particularly susceptible to effects of UV light, and are vitally important to marine food-webs).
An increase of UV radiation would also affect crops. A number of economically important species of plants, such as rice, depend on cyanobacteria residing on their roots for the retention of nitrogen. Cyanobacteria are very sensitive to UV light and they would be affected by its increase.
Aside from the direct effect of ultraviolet radiation on organisms, increased surface UV leads to increased tropospheric ozone, as noted above. Paradoxically, at ground-level ozone is generally recognised to be a health risk, as ozone is toxic due to its strong oxidant properties. At the time, ozone at ground level is produced mainly by the action of UV radiation over combustion gases from vehicle exhausts.
Public policy in response to the ozone hole
Environmentalists assert that the CFCs have caused so much damage to the ozone layer that CFCs should be completely banned. The full extent of the damage CFCs have caused is not known and will not be known for decades; however, marked decreases in column ozone have already been observed (as explained above).
In 1987, the Montreal Protocol was signed, controlling the emissions of CFCs. To some extent, their role has been replaced by the less damaging hydro-chloro-fluoro-carbons (HCFCs), although concerns remain regarding HCFCs also. In some applications, hydro-fluoro-carbons (HFCs) have been used to replace CFCs. HFC's, which contain no chlorine or bromine, do not contribute at all to ozone depletion although they are potent greenhouse gases. The best known of these compounds is probably HFC-134a (R-134a), which in the United States has largely replaced CFC-12 (R-12) in automobile air conditioners.
Pielke and Betsill (http://sciencepolicy.colorado.edu/admin/publication_files/1997.11.pdf) provide an extensive review of early US govt responses to the emerging science of ozone depletion by CFCs.
The future of ozone depletion
Ozone_cfc_trends.png
Since the adoption and strengthening of the Montreal Protocol has led to reductions in the emissions of CFCs, atmospheric concentrations of the most significant compounds have been declining. These substances are being gradually removed from the atmosphere. By 2015, the Antarctic ozone hole should have reduced by only 1 million km2 out of 25 (Newman et al, GRL 2004); complete recovery of the Antarctic ozone layer will not occur until the year 2050 or later.
There is a slight caveat to this, however. Global warming from CO2 is expected to cool the stratosphere. This, in turn, would lead to a relative increase in ozone depletion and the frequency of ozone holes. The effect may not be linear: ozone holes form because of polar stratospheric clouds; the formation of polar stratospheric clouds has a temperature threshold above which they will not form; cooling of the Arctic stratosphere might lead to Antarctic-ozone-hole-like conditions. But at the moment this is not clear.
Even though the stratosphere as a whole is cooling, high-latitude areas may become increasingly predisposed to springtime stratospheric warming events as weather patterns change in response to higher greenhouse gas loading. This would cause PSCs to disappear earlier in the season, and may explain why Antarctic ozone hole seasons have tended to end somewhat earlier since 2000 as compared with the most prolonged ozone holes of the 1990s.
The decrease in ozone-depleting chemicals has also been significantly affected by a decrease in bromine-containing chemicals. The data suggest that substantial natural sources exist for atmospheric methyl bromide (CH3Br).[9] (http://www.wmo.ch/web/arep/reports/ozone_2002/06_chapter1.pdf)
The 2004 ozone hole ended in November 2004, daily minimum stratospheric temperatures in the Antarctic lower stratosphere increased to levels that are too warm for the formation of polar stratospheric clouds (PSCs) about 2 to 3 weeks earlier than in most recent years.[10] (http://www.wmo.ch/web/arep/04/bulletin_7_2004.pdf)
The Arctic winter of 2005 was extremely cold in the stratosphere; PSCs were abundant over many high-latitude areas until dissipated by a big warming event, which started in the upper stratosphere during February and spread throughout the Arctic stratosphere in March. The size of the Arctic area of anomalously low total ozone in 2004-2005 was larger than in any year since 1997. The predominance of anomalously low total ozone values in the Arctic region in the winter of 2004-2005 is attributed to the very low stratospheric temperatures and meteorological conditions favorable for ozone destruction, along with the continued presence of ozone destroying chemicals in the stratosphere.[11] (http://www.cpc.ncep.noaa.gov/products/stratosphere/winter_bulletins/nh_04-05/index.html)
A 2005 IPCC summary of ozone issues observed that observations and model calculations suggest that the global average amount of ozone depletion has now approximately stabilized. Although considerable variability in ozone is expected from year to year, including in polar regions where depletion is largest, the ozone layer is expected to begin to recover in coming decades due to declining ozone-depleting substance concentrations, assuming full compliance with the Montreal Protocol.[12] (http://www.ipcc.ch/press/SPM.pdf)
Controversy regarding ozone science and policy
Any measures against ozone depletion which have a negative economic impact will remain a controversial issue due to the strong economic interests involved, with key questions regarding whether the scientific understanding is strong enough to warrant the proposed countermeasures. In this context, it is worth noting that it is commonly believed that one reason for the relative ease of introduction of the Montreal protocol was the availability of CFC replacements at little extra cost.
The consensus among most atmospheric physicists and chemists is that the scientific understanding has now reached a level where countermeasures to control CFC emissions are justified, although the decision is ultimately one for policy-makers and society.
Some atmospheric scientists (for instance Fred Singer, founder of SEPP) question or completely deny a link between CFCs and ozone depletion [13] (http://www.sepp.org/ozone/ozonefranklin.html).
It is common to see completely nonsensical arguments put forward to prove that CFCs cannot cause ozone depletion — for example, that they are heavier than air and so cannot reach the stratosphere [14] (http://www.junkscience.com/may99/freon.htm). CFCs are heavier than air, but just like argon, krypton and other heavy gases with a long lifetime they are uniformly distributed throughout the turbosphere and reach the upper atmosphere [15] (http://www.so.wustl.edu/science_outreach/curriculum/ozone/info/stratosphere/myths/heavier.html).
Despite this general consensus, the science behind ozone depletion remains complex, and some who oppose the enforcement of countermeasures point to some of the difficulties experienced in these studies. For example, although increased UVB has been shown to constitute a melanoma risk (see above), it has been difficult for statistical studies to establish a direct link between ozone depletion and increased rates of melanoma. Although melanomas did increase significantly during the period 1970-1990, it is difficult to separate reliably the effect of ozone depletion from the effect of changes in lifestyle factors (e.g. increasing rates of foreign travel).
Related articles
- ozone
- ozone layer
- ozone-oxygen cycle
- Montreal Protocol
- Scientific assessment of ozone depletion
- CFC
- melanoma, skin cancer
- greenhouse gas
- ultraviolet
External links
- Stratospheric ozone: an electronic textbook (http://www.ccpo.odu.edu/SEES/ozone/oz_class.htm)
- Scientific Assessment of Ozone Depletion: 2002 (http://www.wmo.ch/web/arep/reports/o3_assess_rep_2002_front_page.html)
- NOAA
- NOAA Stratospheric Ozone Webpage (http://www.ozonelayer.noaa.gov/)
- Stratospheric ozone depletion (Antarctic, Arctic, and global) (http://www.al.noaa.gov/WWWHD/Pubdocs/StratO3.html) at NOAA
- NOAA's Ozone and Water Vapor Group (http://www.cmdl.noaa.gov/ozwv/)
- Scientific Assessment of Ozone Depletion: 1998 (http://www.al.noaa.gov/WWWHD/pubdocs/assessment98.html) - by WMO, UNEP, NOAA
- Ozone Depletion And Global Environmental Change (http://www.ciesin.org/TG/OZ/oz-home.html) at Columbia University
- The Ozone Tour (http://www.atm.ch.cam.ac.uk/tour/) at the Centre for Atmospheric Science, University of Cambridge
- The Ozone FAQ (http://www.faqs.org/faqs/ozone-depletion/) - a superb resource (but last updated in 1997)
- Jon Shanklin's Ozone pages (http://www.antarctica.ac.uk/met/jds/ozone/), including a pamphlet (http://www.antarctica.ac.uk/met/jds/ozone/ozpamw7.htm).
- The Ozone Depletion Story (http://www.nrdc.org/air/pollution/hozone.asp)
- The Nobel Prize in Chemistry 1995 (http://nobelprize.org/chemistry/laureates/1995/index.html) - includes links to biographies of Crutzen, Molina and Rowland and to their Nobel Lectures.
- Ozone Depletion (http://www.ace.mmu.ac.uk/eae/Ozone_Depletion/ozone_depletion.html)
- SEPP
- Five Scientific Questions On The Cfc-Ozone Issue (http://www.sepp.org/ozone/5questions.html) by S. Fred Singer
- A Critique of the UN Scientific Assessment of Ozone Depletion: 1994 (http://www.sepp.org/ozone/ozassm.html) by S. Fred Singer
- The Ozone-CFC Debacle: Hasty Action, Shaky Science (http://www.sepp.org/ozone/ozonefranklin.html) by S. Fred Singer
- The Skeptics vs. the Ozone Hole (http://www.wunderground.com/education/ozone_skeptics.asp)
- Discussion of the ozone layer (http://groups.yahoo.com/group/o3ozone)de:Ozonloch
et:Osooniauk he:החור בשכבת האוזון ja:オゾンホール nl:Ozongat pl:Dziura ozonowa vi:Sự suy giảm ôzôn