X-ray
|
|
- In the NATO phonetic alphabet, X-ray represents the letter X.
An X-ray or Röntgen ray is a form of electromagnetic radiation with a wavelength in the range of 10 nanometers to 100 picometers (corresponding to frequencies in the range 30 PHz to 3 EHz). X-rays are primarily used for diagnostic medical imaging and crystallography. X-rays are a form of ionizing radiation and as such can be dangerous.
| Contents |
Physics
X-rays with a wavelength approximately longer than 0.1 nm are called soft X-rays. At wavelengths shorter than this, they are called hard X-rays. Hard X-rays overlap the range of "long"-wavelength (lower energy) gamma rays, however the distinction between the two terms depends on the source of the radiation, not its wavelength: X-ray photons are generated by energetic electron processes, gamma rays by transitions within atomic nuclei.
The basic production of X-rays is by accelerating electrons in order to collide with a metal target (tungsten usually). Here the electrons suddenly decelerate upon colliding with the metal target and if enough energy is contained within the electron it is able to knock out an electron from the inner shell of the metal atom and as a result electrons from higher energy levels then fill up the vacancy and X-ray photons are emitted.
Detectors
The detection of X-rays is based on various methods. The most commonly known method is the photographic plate, frequently used in hospitals. When a photographic negative plate is exposed to the X-rays, it turns white where the X-rays go through "soft" parts of the body like organs and skin, and black where the X-rays are stopped by "hard" parts like bone, or contrast product containing barium or iodine injected in blood. Another method is to use a fluorescent plate, e.g. sodium iodide NaI. These methods give no information about the energy of the X-ray photons, just their spatial density.
Initially, most common detection methods were based on the ionisation of gases, as in the Geiger-Müller counter: a sealed cylinder with a polymer window contains a gas, and a wire, and a high voltage is applied between the cylinder (cathode) and the wire (anode). When an X-ray photon enters the cylinder, it ionises the gas which becomes conducting, creating a current flow (a kind of flash); this peak of current is detected and is called a "count".
When the high voltage between anode and cathode is decreased, the detector is no longer saturated, and the height of the current peak is proportional to the energy of the photon; it is thus called a "proportional counter". Most of times, the cylinder is not sealed but is constantly fed with "fresh gas", is thus called a "flow counter". This proportionality property allows filtering the "interesting" peaks from the noise and other photons, but the resolution in energy is not enough to determine the energy spectrum; such a feature requires a diffracting crystal to first separate the different photons, the method is called wavelength dispersive X-ray spectroscopy (WDX or WDS).
Some materials such as NaI can "convert" an X photon to a visible photon; an electronic detector can be built by adding a photomultiplier. These detectors are called "scintillators" or "scintillation counters".
Since the 1970s, new semiconductor diode detectors have been developed (silicon or germanium doped with lithium, Si(Li) or Ge(Li)). X-ray photons are converted to electron-hole pairs in the semiconductor, and are collected to detect the X-rays. When the temperature is low enough (the detector is cooled by Peltier effect or best by liquid nitrogen), it is possible to directly determine the X-ray energy spectrum; this method is called energy dispersive X-ray spectroscopy (EDX or EDS); it is often used in small X-ray fluorescence spectrometers. These detectors are often called "solid detectors".
It is commonly thought that X-rays are invisible to the human eye, and for almost all everyday uses of X-rays this may seem true, however, very strictly speaking, it is actually false. In special circumstances, X-rays are in fact visible to the "naked eye". An effect first discovered by Brandes in experimentation a short time after Röntgen's landmark 1895 paper; he reported, after dark adaptation and placing his eye close to an X-ray tube, seeing a faint "blue-gray" glow which seemed to originate within the eye itself.[1] (http://www.orau.org/ptp/articlesstories/invisiblelight.htm) Upon hearing this, Röntgen reviewed his record books and found he in fact, also saw the effect. When placing an X-ray tube on the opposite side of a wooden door Röntgen saw the same blue glow seeming to emanate from the eye itself, but thought his observations were spurious due to the fact that he only saw the effect when he used one type of tube. Later he realized that the tube which created the effect was the only one which produced X-rays powerful enough to make the glow plainly visible and the experiment was thereafter repeated readily. The fact that X-rays are actually faintly visible to the dark-adapted naked eye has largely been forgotten today is probably due to the lack of desire to repeat what we would now see as a recklessly dangerous and harmful experiment with ionizing radiation. It is not known what the exact mechanism in the eye is which produces the visibility and it could be due to either conventional detection (excitation of rhodopsin molecules in the retina), direct excitation of retinal nerve cells, or secondary detection via, for instance, X-ray induction of phosphorescence in the eyeball and then conventional retinal detection of the secondarily produced visible light.
Medical uses
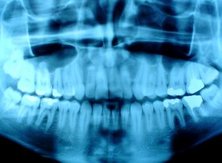
Since Röntgen's discovery that X-rays can identify bony structures, X-rays have been developed for their use in medical imaging. Radiology is a specialised field of medicine that employs radiography and other techniques for diagnostic imaging. Indeed, this is probably the most common use of X-ray technology.
The use of X-rays are especially useful in the detection of pathology of the skeletal system, but are also useful for detecting some disease processes in soft tissue. Some notable examples are the very common chest X-ray, which can be used to identify lung diseases such as pneumonia, lung cancer or pulmonary oedema, and the abdominal X-ray, which can detect ileus (blockage of the intestine), free air (from visceral perforations) and free fluid (in ascites). In some cases, the use of X-rays is debatable, such as gallstones (which are rarely radiopaque) or kidney stones (which are often visible, but not always). Also, Traditional plain X-rays pose very little use in the imaging of soft tissues such as the brain or muscle. Imaging alternatives for soft tissues are computed axial tomography (CAT or CT scanning), magnetic resonance imaging (MRI) or ultrasound.
X-rays are also used in "real-time" procedures such as angiography or contrast studies of the hollow organs (e.g. barium enema of the small or large intestine) using fluoroscopy. Angioplasty, medical interventions of the arterial system, rely heavily on X-ray-sensitive contrast to identify potentially treatable lesions.
Radiotherapy, a curative medical intervention, now used almost exclusively for cancer, employs higher energies of radiation.
History
Among the important early researchers in X-rays were Professor Ivan Pului, Sir William Crookes, Johann Wilhelm Hittorf, Eugene Goldstein, Heinrich Hertz, Philipp Lenard, Hermann von Helmholtz, Nikola Tesla, Thomas Edison, Charles Glover Barkla, Max von Laue, and Wilhelm Conrad Röntgen.
Roentgen-Roehre.png
Physicist Johann Hittorf (1824 - 1914) observed tubes with energy rays extending from a negative electrode. These rays produced a fluorescence when they hit the glass walls of the tubes. In 1876 the effect was named "cathode rays" by Eugene Goldstein. Later, English physicist William Crookes investigated the effects of energy discharges on rare gases, and constructed what is called the Crookes tube. It is a glass vacuum cylinder, containing electrodes for discharges of a high voltage electric current. He found, when he placed unexposed photographic plates near the tube, that some of them were flawed by shadows, though he did not investigate this effect.
In April 1887, Nikola Tesla began to investigate X-rays using high voltages and vacuum tubes of his own design, as well as Crookes tubes. From his technical publications, it is indicated that he invented and developed a special single-electrode X-ray tube, which differed from other X-ray tubes in having no target electrode. He stated these facts in his 1897 X-ray lecture before the New York Academy of Sciences.
The principle behind Tesla's device is nowadays called the bremsstrahlung process, in which a high-energy secondary X-ray emission is produced when charged particles (such as electrons) pass through matter. By 1892, Tesla performed several such experiments, but he did not categorize the emissions as what were later called X-rays, instead generalizing the phenomenon as radiant energy. He did not publicly declare his findings nor did he make them widely known. His subsequent X-ray experimentation by vacuum high field emissions led him to alert the scientific community to the biological hazards associated with X-ray exposure.
In 1892, Heinrich Hertz began experimenting and demonstrated that cathode rays could penetrate very thin metal foil (such as aluminium). Philipp Lenard, a student of Heinrich Hertz, further researched this effect. He developed a version of the cathode tube and studied the penetration by X-rays of various materials. Philipp Lenard, though, did not realize that he was producing X-rays. Hermann von Helmholtz formulated mathematical equations for X-rays. He postulated a dispersion theory before Röntgen made his discovery and announcement. It was formed on the basis of the electromagnetic theory of light (Wiedmann's Annalen, Vol. XLVIII). However, he did not work with actual X-rays.
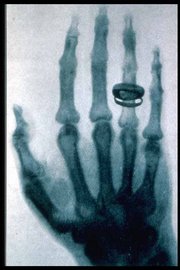
On November 8 1895, Wilhelm Röntgen, a German scientist, began observing and further documenting X-rays while experimenting with vacuum tubes. Röntgen, on December 28, 1895, wrote a preliminary report "On a new kind of ray: A preliminary communication". He submitted it to the Würzburg's Physical-Medical Society journal. This was the first formal and public recognition of the categorization of X-rays. Röntgen referred to the radiation as "X", to indicate that it was an unknown type of radiation. The name stuck, although (over Röntgen's great objections), many of his colleagues suggested calling them Röntgen rays. They are still referred to as such in many languages, see "in other languages" in the left margin of this article. Röntgen received the first Nobel Prize in Physics for his discovery.
In 1895, Thomas Edison investigated materials' ability to fluoresce when exposed to X-rays, and found that calcium tungstate was the most effective substance. Around March 1896, the fluoroscope he developed became the standard for medical X-ray examinations. Nevertheless, Edison dropped X-ray research around 1903 after the death of Clarence Madison Dally, one of his glassblowers. Dally had a habit of testing X-ray tubes on his hands, and acquired a cancer in them so tenacious that both arms were amputated in a futile attempt to save his life[2] (http://www.ratical.org/radiation/KillingOurOwn/KOO6.html). In 1906, physicist Charles Barkla discovered that X-rays could be scattered by gases, and that each element had a characteristic X-ray. He won the 1917 Nobel Prize in Physics for this discovery.
The use of X-rays for medical purposes (to develop into the field of radiation therapy) was pioneered by Major John Hall-Edwards in Birmingham, England. In 1908, he had to have his left arm amputated owing to the spread of X-ray dermatitis[3] (http://www.birmingham.gov.uk/xray). In the 1950s X-rays were first harnessed to produce an X-ray microscope.
Moon_in_x-rays.gif
In the 1980s an X-ray laser device was proposed as part of the Reagan administration's Strategic Defense Initiative, but the first and only test of the device (a sort of laser "blaster", or death ray, powered by a thermonuclear explosion) gave inconclusive results. For technical and political reasons, the overall project (including the X-ray laser) was de-funded (though was later revived by the second Bush administration as National Missile Defense using different technologies).
In the 1990s the Chandra X-Ray Observatory (http://chandra.harvard.edu/) was launched, allowing the exploration of the very violent processes in the universe which produce X-Rays. Unlike visible light, which is a relatively stable view of the universe, the x-ray universe is unstable, it features stars being torn apart by black holes, galactic collisions, and novas, neutron stars that build up layers of plasma that then explode into space.
See also
| Electromagnetic Spectrum
Radio waves | Microwave | Terahertz radiation | Infrared | Optical spectrum | Ultraviolet | X-ray | Gamma ray Visible: Red | Orange | Yellow | Green | Blue | Indigo | Violet |
de:Röntgenstrahlen es:Rayos X fr:Rayon X ko:X선 id:Sinar-X it:Raggi X ms:Sinar-X nl:Röntgenstraling ja:X線 no:Røntgenstråling pl:Promieniowanie rentgenowskie pt:Raios X sv:Röntgenstrålning zh:X射线