Atom
|
|
| Atom | ||||||
|---|---|---|---|---|---|---|
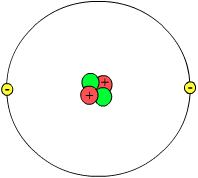
| ||||||
| Helium atom model (not to scale) Showing nucleus with two protons (red) and two neutrons (green) and with a probability cloud (gray) of two electrons (yellow). | ||||||
| Classification | ||||||
| ||||||
| Properties | ||||||
|
An atom is a microscopic structure found in all ordinary matter around us.
Atoms are composed of 3 types of subatomic particles:
- electrons, which have a negative charge;
- protons, which have a positive charge; and
- neutrons, which have no charge.
Atoms are the fundamental building blocks of chemistry, and are conserved in chemical reactions. An atom is the smallest particle differentiable as a certain chemical element. Only 90 elements (of 112 now known) have been identified as occurring naturally on Earth; the rest can be created in the lab, and some (for example, technetium and californium have been identified in supernovae.
Each element is unique by the number of protons in each atom of that element. Every neutral atom has a number of electrons equal to its number of protons; if there is an imbalance, the atom has an electric charge and is called an ion. Atoms of the same element can have different numbers of neutrons, as long as the number of protons does not change. Atoms with different numbers of neutrons (but the same number of protons) are called isotopes of a chemical element.
Atoms are able to bond into molecules and other types of chemical compounds. Molecules are made up of multiple atoms; For example, a molecule of water is a combination of 2 hydrogen and one oxygen atom.
Because of their ubiquitous nature, atoms have been an important field of study for many centuries. Current research focuses on quantum effects, such as in Bose-Einstein condensate.
| Contents |
Atomic theory
The Atomic theory is a theory of the nature of matter. It states that all matter is composed of atoms.
Atom sizes
The size of an atom is not easily defined since the electron orbitals just gradually go to zero as the distance from the nucleus increases. For atoms that can form solid crystals, the distance between adjacent nuclei can give an estimate of the atom size. For atoms that do not form solid crystals other techniques are used, including theoretical calculations. As an example, the size of a hydrogen atom is estimated to be approximately 1.2×10-10m. Compare this to the size of the proton which is the only particle in the nucleus of the hydrogen atom which is approximately 0.87×10-15m. Thus the ratio between the sizes of the hydrogen atom to its nucleus is about 100,000. Atoms of different elements do vary in size, but the sizes are roughly the same to within a factor of 2 or so. The reason for this is that elements with a large positive charge on the nucleus attract the electrons to the center of the atom more strongly.
Elements and isotopes
Atoms are generally classified by their atomic number, which corresponds to the number of protons in the atom. The atomic number defines which element the atom is. For example, carbon atoms are those atoms containing 6 protons. All atoms with the same atomic number share a wide variety of physical properties and exhibit the same chemical behavior. The various kinds of atoms are listed in the periodic table in order of increasing atomic number.
The mass number, atomic mass number, or nucleon number of an element is the total number of protons and neutrons in an atom of that element, because each proton or neutron essentially has a mass of 1 amu. The number of neutrons in an atom has no effect on which element it is. Each element can have numerous different atoms with the same number of protons and electrons, but varying numbers of neutrons. Each has the same atomic number but a different mass number. These are called the isotopes of an element. When writing the name of an isotope, the element name is followed by the mass number. For example, carbon-14 contains 6 protons and 8 neutrons in each atom, for a total mass number of 14.
The simplest atom is the hydrogen atom, which has atomic number 1 and consists of one proton and one electron. The hydrogen isotope which also contains 1 neutron is called deuterium or hydrogen-2; the hydrogen isotope with 2 neutrons is called tritium or hydrogen-3.
The atomic mass listed for each element in the periodic table is an average of the isotope masses found in nature, weighted by their abundance.
Valence and bonding
The chemical behavior of atoms is largely due to interactions between electrons. Electrons of an atom must remain within certain, predictable electron configurations. Electrons fall into shells based on their relative energy level which is usually visualized as distance from the nucleus. The electrons in the outermost shell, called the valence electrons, have the greatest influence on chemical behavior. Core electrons (those not in the outer shell) play a role, but it is usually in terms of a secondary effect due to screening of the positive charge in the atomic nucleus.
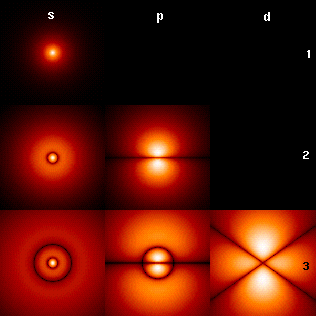
Each shell, numbered from the one closest to the nucleus (lowest in energy), can hold up to a specific number of electrons due to its differing sublevel and orbital capacity:
- Shell 1: 2 electron capacity - s sublevel - 1 orbital
- Shell 2: 8 electron capacity - s and p sublevels - 4 orbitals
- Shell 3: 18 electron capacity - s, p, and d sublevels - 9 orbitals
- Shell 4: 32 electron capacity - s, p, d, and f sublevels - 16 orbitals
To determine the electron capacity of a shell, the formula 2n2 is used, when n = shell # or principle quantum number. Electrons fill orbitals and shells from the inside out, beginning with shell one. Whichever occupied shell is currently most outward is the valence shell, even if it only has one electron.
The reason why shells fill up in order is that the energy levels of electrons in the innermost shells are significantly lower than the energy levels of electrons in outer shells. So if the inner shells were not completely full, the electron in an outer shell would quickly "fall" into the inner shell (with the emission of a photon that would carry away the difference in the energy.
The number of electrons in an atom's outermost valence shell governs its bonding behavior. Therefore, elements with the same number of valence electrons are grouped together in the periodic table of the elements. Group (i.e. column) 1 elements contain one electron on their outer shell; Group 2, two electrons; Group 3, three electrons; etc. As a general rule, the fewer electrons in an atom's valence shell, the more reactive it is. Group 1 metals are therefore very reactive, with caesium, rubidium, and francium being the most reactive of all metals.
Every atom is much more stable (i.e. less energetic) with a full valence shell. This can be achieved one of two ways: an atom can either share electrons with neighboring atoms (a covalent bond), or it can remove electrons from other atoms (an ionic bond). Another form of ionic bonding involves an atom giving some of its electrons to another atom; this also works because it can end up with a full valence by giving up its entire outer shell. By moving electrons, the two atoms become linked. This is known as chemical bonding and serves to build atoms into molecules or ionic compounds. Five major types of bonds exist:
Atoms in the universe and our world
Using inflation theory, the number of atoms in the observable universe can be estimated to be between 4×1078 and 6×1079. However, because of the possibly infinite nature of the universe, the total number of atoms in the entire universe may be much larger or even infinite. This does not change the estimated number of atoms in the observable universe since that is the number of atoms within about 14 billion light years of us - which is all that we can observe since the universe is only about 14 billion years old.
Atoms in industry
Atoms serve many important roles in industry, including in nuclear power plants, industrial materials science, and many roles in the chemical industry.
Atoms in science
The study of atoms has been a major focus of scientific research for decades. Atomic theory has wide-ranging impact on many fields of science, including nuclear physics, spectroscopy, and all of chemistry, among many other topics. Today, most scientific research associated with atoms is in the field of quantum mechanics. Subatomic particle research is also a popular field.
The study of atoms was done by largely indirect means through the 19th century and early 20th century. In recent years, however, new techniques have made the identification and study of atoms easier and more accurate. The electron microscope, invented in 1931, has allowed pictures to be taken of actual, individual atoms. Methods also exist to identify atoms and compounds. Mass spectrometry methods allow the exact identification of the types and amounts of atoms in a substance. GCMS is often used by forensics investigators to identify unknown substances. X-ray crystallography reveals the structure of certain atomic and molecular substances.
History
Historical theories
Democritus and Leucippus, Greek philosophers in the 5th century BC, presented the first theory of atoms. They held that each atom had a different shape, like a pebble, that governed the atom's properties. Dalton's work in the 19th century proved that matter was made up of atoms, but he knew nothing of their structure. This goes against the theory of infinite divisibility, which states that matter can always be divided into smaller parts.
Through this time, atoms were thought to be the smallest possible piece of matter. However, it was later shown that atoms are made up of subatomic particles. Thomson's experiments discovered the electron, the first of the subatomic particles to be discovered. This showed that atoms are actually divisible. Rutherford's work helped to show that the positively charged nucleus exists. All recent models of the atom have taken into account the existence of subatomic particles.
Since Democritus' time, many theories for the structure of the atom have been suggested, including:
- Plum pudding model,
- Cubical atom,
- Bohr model, and
- the quantum theory of atoms, which is currently accepted and discussed above
While Democritus's theory has been widely disproven, the recent string theory is based on a similar idea of shape and vibration governing a subatomic particle's properties.
Etymology of "atom"
The word "atom" is derived from the Greek atomos, "indivisible", from a-, not, and tomos, a cut. Until the discovery of the electron in the late 19th century, it was believed that atoms were tiny, indivisible particles. Template:Chem clipart
